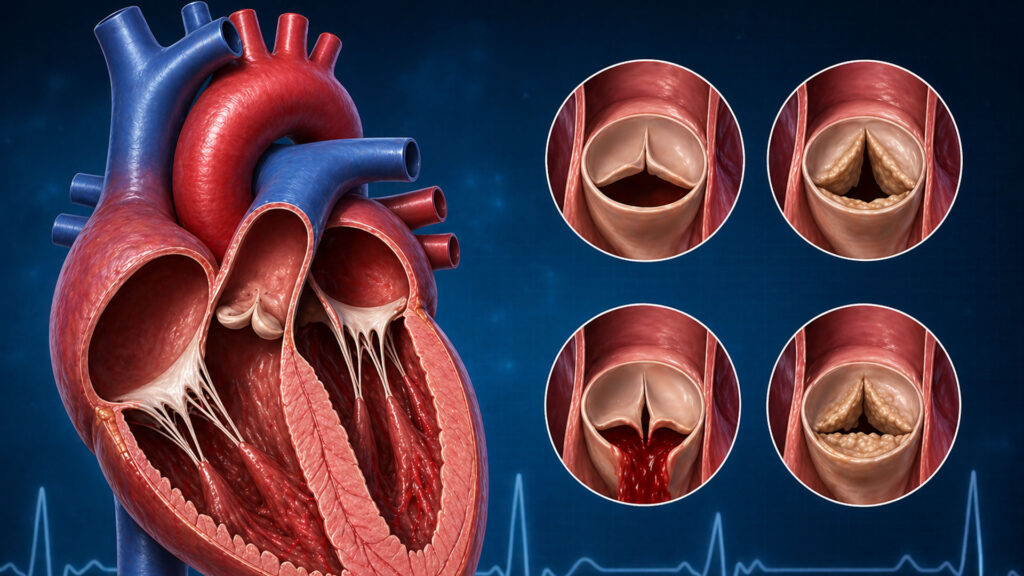
Definition of Heart Failure
Heart support devices (HSD) are high-technology devices produced to take over the pumping duty of the heart in patients with end-stage heart failure. There are models whose mechanical parts can be placed inside the chest or abdomen, as well as paracorporal (outside the body) models. Generally, the battery is located outside the body. The fundamental reason leading to the development of these devices is that some of the patients waiting on the heart transplant candidate list are lost during the waiting period due to limitations in finding donors. There have been major developments regarding HSDs in the last 10 years, and significant progress has been made toward reaching ideal devices. While the 1-year survival rate was 50% in the first-generation artificial support devices, this rate was reported as 68-86% in the 2nd generation devices and around 90% in the 3rd generation devices. Following the successful results obtained with systems used in the treatment of cardiogenic shock in the early years, multi-purpose use is increasing today with the aims of keeping the patient alive until transplantation (bridge to transplantation) and improving heart failure (bridge to recovery). In the REMATCH study concluded in the USA in 2002, the superiority of implantable HSDs over optimal medical treatment in terms of survival and quality of life in patients not suitable for heart transplantation resulted in the granting of FDA approval for long-term use (destination therapy). In patient selection, besides updated indications, the cardiac and extracardiac characteristics of the patient are the primary determinants, while in device and system selection, preference for the one most suitable for the patient’s clinical picture is essential. In recent years, along with well-known pulsatile systems, interest in miniaturized flow pumps has been increasing. Consequently, it is suggested that in the next decade, the transformation of HSDs into a special field of expertise within heart failure surgery, new organizations inside and outside hospitals, and financing regulations in the health system will necessitate a total institutional restructuring in societies.
Heart transplantation and artificial heart support devices are performed by fully equipped hospitals authorized by the R.T. Ministry of Health, which have experienced teams in organ transplantation and artificial heart systems and carry out heart transplantation programs.
Who Needs an Auxiliary Artificial Heart Device and When?
Auxiliary heart support devices are mainly used for the following purposes in patients with end-stage heart failure:
- For short-term use (short-term applied devices) in patients who suddenly develop heart failure, to prevent deterioration in other organ functions and to keep the patient alive until a long-term support system is applied,
- For the purpose of recovery (short-term applied devices) in diseases that cause sudden heart failure such as inflammation of the heart muscle (Myocarditis) but heal completely as a result of drug treatment, in order to keep the patient alive during the severe period of the disease,
- To keep the patient alive until heart transplantation (long-term applied devices) in patients who are on the waiting list for heart transplantation but whose heart failure progresses while waiting for a heart,
- For lifelong use (long-term applied devices) to increase the life span and quality of patients who cannot undergo heart transplantation for various reasons (advanced age, serious kidney, liver disease, etc.),
- To provide the chance of heart transplantation later (long-term applied devices) in patients who cannot undergo heart transplantation due to high pulmonary (lung) resistance/pressure (PVR), as a decrease in PVR can be seen after VAD implantation.
If severe failure has developed in both the right and left sides of the heart, a total artificial heart device is placed.
Total Artificial Heart Device
It is the device that is placed in stead of the diseased heart after it is removed from the body and carries out the circulation. It keeps the patient alive for temporary purposes, that is, until heart transplantation.
Frequently Asked Questions and Problems Regarding Auxiliary Artificial Heart Devices
Artificial heart support devices are in two different groups: those placed inside the body (implantable, miniature) and those outside the body (paracorporal). Which one will be installed is determined according to the characteristics of the patient’s heart failure and general condition. Although the artificial heart support device surgery varies according to the type of device installed, if no complications develop, it is a surgery that lasts 4-6 hours on average. The pump stays inside your chest and is directly connected to your heart. It pumps blood from the left side of your heart into your aorta (the large blood vessel that carries blood from your heart to other parts of your body). Patients whose surgery is finished wake up in a special transplant room in the intensive care unit; they are disconnected from the respiratory device when all findings return to normal. Blood comes out of the pump in a continuous flow, like water coming out of a garden hose. This continuous flow is different from the pulse of a normal heart and as a result, you may not be able to feel your pulse. There is no need to worry, this is a normal situation. These patients have to use blood-thinning medications for as long as they live with the device and consult their doctors for the doses of these medications by having regular blood tests. The pump works with a small external computer called a controller. The pump and controller are connected to each other by a small cable (driveline) passing through the skin in your upper abdominal region. The controller runs the pump and also provides text messages and audible alarms for you to check the operation of the system. The controller works with two batteries or one battery and wall electricity. The entire system is portable and weighs approximately 1.5 kg. You can attach a carrying case to your waist or carry it on your shoulder, so you take the system everywhere you go.
Artificial Heart Device Program
The Artificial Heart Support Device Program at our hospital began in 1990 and has continued to the present day. In 2001, the first intracardiac heart pump in Türkiye was successfully implanted in our patient. Technological developments regarding artificial heart support devices are closely monitored by our teams. Today, miniaturized heart pumps used in leading transplant centers worldwide are also utilized for our patients at our hospital. In 2013, for the first time at our hospital, total artificial heart devices were successfully implanted in two of our patients.
Share